
Capability Highlights
- ISO 13485 Compliant R2E™ Design Controls
- ISO 13485 Transition to Manufacturing as a Service
- ISO 14971 Risk Assessments and Management
- IEC 62304 Medical Device Software Lifecycle Processes
- IEC 60601 / IEC 61010-1 Medical Electrical Equipment Safety
- IEC 61000 Electromagnetic Compatibility (EMC)
Industry Summary
Marcus Engineering has deep experience with medical electronics and medical device development. Our team works collaboratively with you to define requirements, user experience, and critical safety considerations, simultaneously developing the comprehensive technical construction file required to pass FDA scrutiny. We are your dedicated partner in navigating the complex path to both functional and regulatory success.
Deeper Dive
Navigating the Regulatory Landscape
Medical device development is a rigorous engineering process that requires strict adherence to FDA standards, extending far beyond the typical design-prototype-deliver cycle. Success demands a foundation of objective evidence and meticulous traceability between customer requirements, risk mitigations, and final design outputs. Whether your product is Class I, II, or III and requires a Premarket Notification (510k), a full Premarket Approval (PMA), or a De Novo application, our team has the expertise to guide your product through the appropriate regulatory pathway toward a successful market launch.
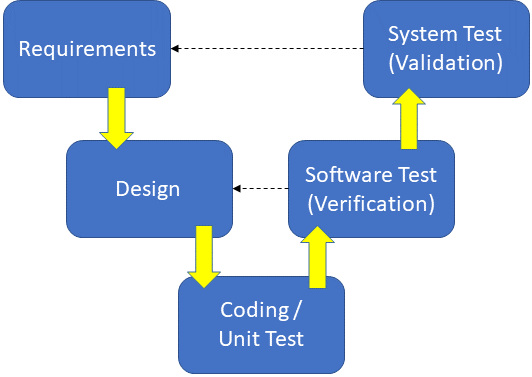
R2E™: A Disciplined Framework for Medical Innovation
At the core of our process is R2E™ (Risk Reduction Engineering™), our disciplined and efficient framework for medical device design controls. Fundamentally, R2E™ is a methodology focused on minimizing risk, maximizing clarity, and addressing foundational design and requirements issues early in the development process while they are still manageable. This proactive approach ensures that not only are functional, performance, and customer requirements met, but that all regulatory and safety requirements are holistically addressed and documented from day one. The R2E™ design approach drastically increases on-time delivery, enhances transparency, and ensures your project is built on a solid, compliant foundation.
Our sophisticated and integrated hardware and software development processes are built upon this R2E™ framework, ensuring every aspect of your device is developed with the highest level of discipline and control.
Medical Device Projects
Projects under NDA. Please contact us!

